New test kits give answers within 45 minutes

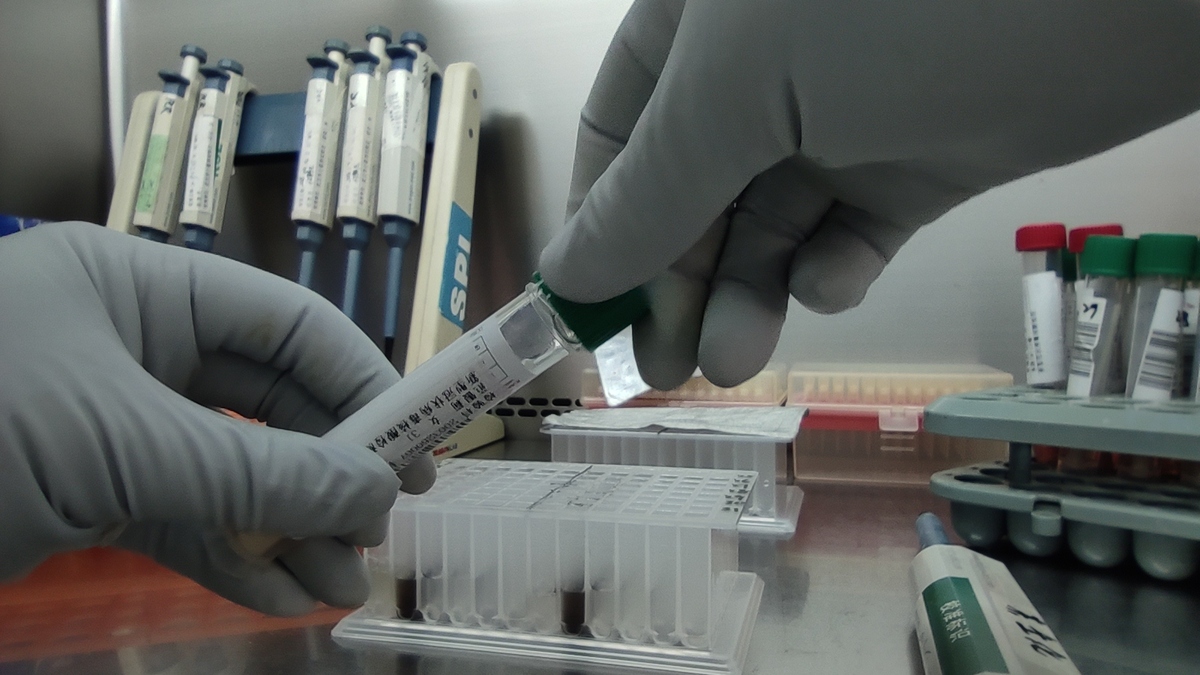
According to the China Association for In-Vitro Diagnostics, laboratory testing for the novel coronavirus is highly dangerous. Employees must wear protective gear that meets biological safety level 3 requirements, the third-highest on a four-tier system for handling dangerous pathogens such as yellow fever, West Nile virus and bacteria that cause tuberculosis.
Zhang said: "To protect the testing staff, the samples are often sterilized for at least 30 minutes at 56 C. This procedure may destroy the virus' genetic material, making it difficult to detect the pathogen."
To solve these problems, China has optimized its biosafety standards, reducing the need to unduly process the samples, she said. Dozens of advanced bioresearch facilities nationwide have also been mobilized to serve as test centers for the virus.
In addition, Wang Jianbin said many designated hospitals have set up test labs, avoiding the need to transport samples to another testing facility, risking contamination in the process.
As the pandemic virus is just a single strand of fragile ribonucleic acid, samples that are collected must be tested within four hours to prevent the genetic material from degrading. If, for some reason, samples are not tested within 24 hours, they must be stored at a temperature of below minus 70 C, according to the China Association for In-Vitro Diagnostics.